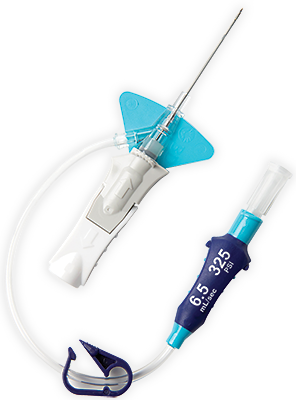
Surmodics (Nasdaq:SRDX) announced today that it received formal feedback from the FDA related to its SurVeil drug-coated balloon (DCB). SRDX shares slipped -1.4% to $18.48 apiece in mid-afternoon trading.įiled Under: Catheters, Mergers & Acquisitions Tagged With: NorMedix, SurModics Inc. Groshong Central Venous Catheters, Groshong NXT ClearVue Peripherally Inserted. Ancora Heart, Egnite partner on AccuCinch trial enrollment. The company raised its revenue outlook from between $60 and $64 million to between $62 million to $66 million. Available in five lengths from 65 to 200 cm and compatible with 0.014, 0.018, and 0.035 guidewires, the Sublime microcatheters feature Surmodics’ best-in-class Pristyne hydrophilic coating for. “NorMedix has been focused on addressing challenging healthcare problems with pioneering design and development capabilities and is a strong fit with SurModics’ culture, capabilities and commitment to providing innovative healthcare solutions to help patients.”Įden Prairie, Minn.-based SurModics, which saw its share price take a hit after it lowered its guidance due to the Creagh buyout, lowered its as-reported earnings outlook from 38¢ to 43¢ per share to between 30¢ and 35¢ per share (adjusted EPS are still pegged at 66¢ to 75¢). Our ReVene Venous Thrombectomy Catheter, which received FDA 510(k) clearance in fiscal 2021, is a mechanical thrombectomy catheter for use in venous vascular beds that is specifically designed to remove large, mixed-morphology blood clots commonly found with VTE. “We are pleased to be joining the SurModics team, recognizing the strategic fit of our design and development expertise with SurModics’ innovative coating technologies,”added NorMedix president & CEO Gregg Sutton, who’s slated to join SurModics as R&D vice president. Looking at the SRDX split history from start to finish, an original position size of 1000 shares would have turned into 2000 today. “We remain focused on being a strategic partner to industry-leading medical device companies by offering early access to innovative whole-product solutions.” “With NorMedix we will have a differentiated catheter-technology platform and additional design and development expertise that will enhance the value we offer our medical device customers,” president & CEO Gary Maharaj said in prepared remarks. Title: Sublime Radial Access 014 Case Report (Dr. Federa a restricts this deice to sae or on the order o a phsician or proper icensed practitioner. Plymouth, Minn.-based NorMedix makes thin-walled catheters, according to SurModics, which inked a $32 million deal for Creagh Medical late last year. The ReVene Thrombectomy Catheter has the potential to significantly expand the use and accessibility of venous mechanical thrombectomy by allowing physicians to intervene early and complete the procedure in a single session, said Stephen Black, principal investigator and leading enroller of the VETEX feasibility study and Consultant. The IVUS catheter was removed and a Sublime 2.0 mm x 220 mm balloon was inserted, advanced to the pedal loop and inflated for two minutes (Figure 2). SurModics (NSDQ: SRDX) said today that it acquired NorMedix for $14 million, including $7 million in up-front cash and another $7 million in revenue milestones.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
